ExiPrep™ 48 Viral DNA/RNA Kit is designed for accurate and rapid extraction of viral DNA or RNA from various human specimens using ExiPrep™ 48 Dx .For HBV and HCV detection, a single sample can be used to detect both targets. The entire extraction process is carried out automatically via ExiPrep™ 48 Dx instrument.
| Weight | 3.25 kg |
|---|---|
| Certification | CE |
| Incoterms | EXW |
$432.00
Features
1. Pre-filled buffer cartridge system
2. Simultaneous extraction of nucleic acids from maximum of 48 clinical samples
3. ExiPrep™48 Dx system uses silica magnetic beads developed and produced with Bioneer’s technology.
Specification
|
Technology |
Magnetic Silica Beads |
||
|
Main sample type |
Body fluids, Swab, Sputum |
||
|
Starting volume |
Serum |
800 ul/400 ul |
|
|
Plasma |
|||
|
Urine |
|||
|
Whole blood |
200 ul |
||
|
Elution volume |
50 ul |
||
|
Instrumention |
ExiPrep™48 Dx |
||
|
Test |
96 |
||
|
Certification |
CE, MFDS |
||
Workflow
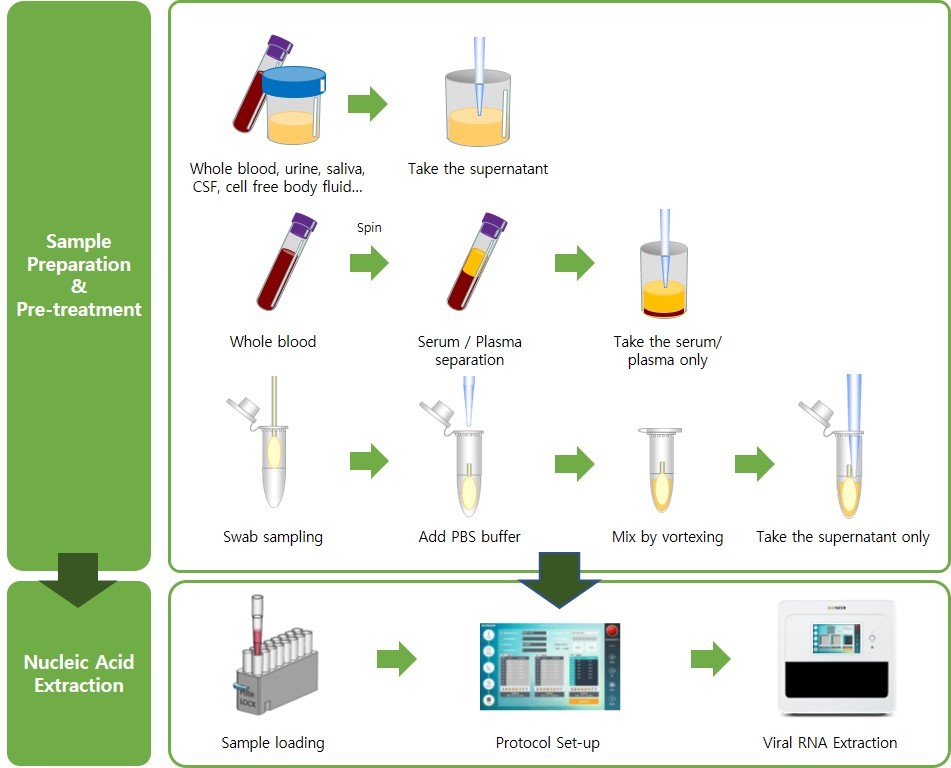
Performances
1.Test results using ExiPrep™ 48 Viral DNA/RNA Kit and AccuPower® HBV Quantitative PCR Kit
|
HBV |
1.64 Log10 IU/ml (N=9) |
3.33 Log10 IU/ml (N=9) |
|
Parameter |
Log10 IU/ml |
Log10 IU/ml |
|
Avg(range) |
1.81(1.62-1.99) |
3.32(3.18-3.42) |
|
SD |
0.13 |
0.09 |
|
%CV |
6.98% |
2.66% |
|
Bias* |
0.17 |
0.01 |
The efficiency of ExiPrep™ 48 Viral DNA/RNA Kit was confirmed with two quantitative concentrations of HBV panel. Viral DNA was extracted from serum using ExiPrep™ Viral DNA/RNA Kit and real-time PCR was performed with AccuPower® HBV Quantitative PCR Kit.
2. Test results using ExiPrep™ 48 Viral DNA/RNA Kit and AccuPower® HCV Quantitative PCR Kit.
|
HCV |
1.48 Log10 IU/ml (N=9) |
3 Log10 IU/ml (N=9) |
|
Parameter |
Log10 IU/ml |
Log10 IU/ml |
|
Avg(range) |
1.92(1.71-2.04) |
2.91(2.84-2.97) |
|
SD |
0.09 |
0.04 |
|
%CV |
4.90% |
1.48% |
|
Bias* |
0.44 |
0.09 |
The efficiency of ExiPrep™ 48 Viral DNA/RNA Kit was confirmed with two quantitative concentrations of HCV panel. Viral RNA was extracted from plasma using ExiPrep™ Viral DNA/RNA Kit and real-time PCR was performed with AccuPower® HCV Quantitive PCR Kit.
3. Test results using ExiPrep™ 48 Viral DNA/RNA Kit and AccuPower® CMV Quantitative PCR Kit
|
CMV |
2.38 Log10 IU/ml (N=20) |
3 Log10 IU/ml (N=20) |
|
Parameter |
Log10 IU/ml |
Log10 IU/ml |
|
Avg(range) |
2.41(2.33-2.55) |
3.00(2.94-3.11) |
|
SD |
0.07 |
0.05 |
|
%CV |
2.95% |
1.67% |
|
Bias* |
0.03 |
0 |
The efficiency of ExiPrep™ 48 Viral DNA/RNA Kit was confirmed with two quantitative concentrations of CMV panel. Viral DNA was extracted from whole blood using ExiPrep™ Viral DNA/RNA Kit and real-time PCR was performed with AccuPower® CMV Quantitative PCR Kit.
Additional information
| Weight | 3.25 kg |
|---|---|
| Certification | CE |
| Incoterms | EXW |
Only logged in customers who have purchased this product may leave a review.
Vendor Information
- Store Name: BIONEER Corporation
- Vendor: BIONEER Corporation
-
Address:
8-11, Munpyeongseo-ro
Daejeon
34302
South Korea - No ratings found yet!






Reviews
There are no reviews yet.