- MOQ: 10,000 Tests
- NMPA Certified factory, CE certified
| Minimum Order | 100 Units |
|---|
$2.50
4360272 in stock
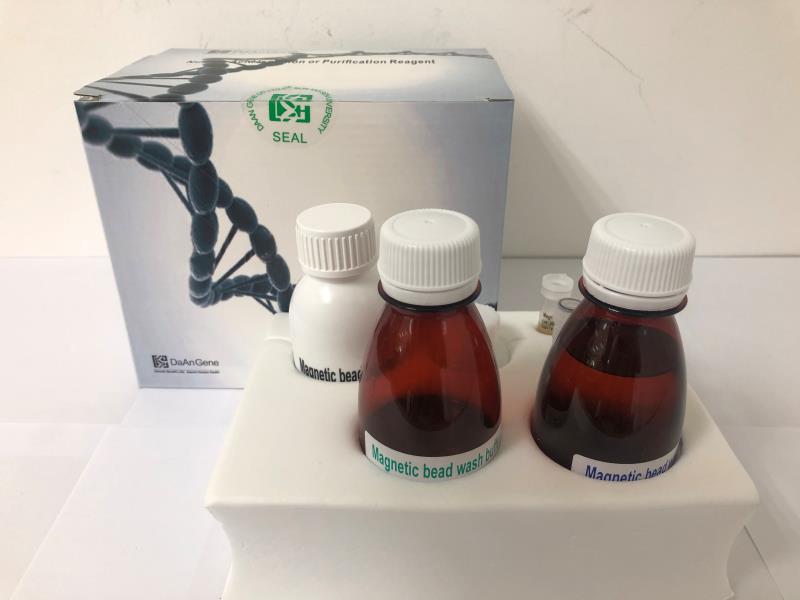
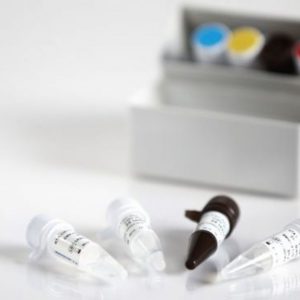
This kit is used for the in vitro qualitative detection of novel coronavirus (2019-nCoV) ORFlab and N gene in the throat swabs, sputum specimens of suspected pneumonia patients infected by a novel coronavirus, suspected clustering cases, and others needing a diagnosis or differential diagnosis for novel coronavirus. For the definitions of "suspected cases" and "suspected clustering cases", refer to the documents(current versions) such as Diagnosis and Treatment Scheme for Pneumonia Patients Infected by NovelCoronavirus and Monitoring Scheme for Pneumonia Patients Infected by Novel Coronavirus issued by the Chinese Center for Disease Control and Prevention (China CDC). This product is only used for the auxiliary diagnosis and in vitro diagnosis (IVD) of pneumonia infected by a novel coronavirus (2019-nCoV) since December 2019 under emergency. Instead, it cannot be clinically used as a general IVD reagent. In use, the relevant requirements of the documents such as Diagnosis and Treatment Scheme for Pneumonia Patients Infected by Novel Coronavirus and Monitoring Scheme for Pneumonia Patients Infected by Novel Coronavirus shall be followed. Detection of novel coronavirus RNA shall meet the requirements of the Technical Guideline of the China CDC for Laboratory Detection of New Coronavirus Pneumonia and other documents, so as to carry out the regulations on biosafety. The detection results of this kit are for clinical reference only and should not be used as the sole criteria for clinical diagnosis. It is recommended to conduct a comprehensive analysis of the condition in combination with the clinical manifestations of the patient and other laboratory tests.
Additional information
| Minimum Order | 100 Units |
|---|
Only logged in customers who have purchased this product may leave a review.
Vendor Information
- Store Name: Sinopharm Fortune Way Company
- Vendor: Sinopharm Fortune Way Company
- Address: China
- No ratings found yet!



Reviews
There are no reviews yet.