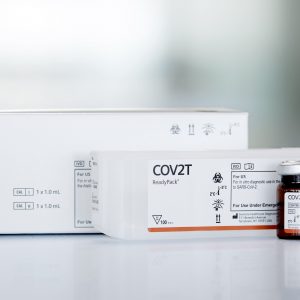
- Intended Use: An aid to determine if individuals may have been exposed and infected by this virus if they have mounted a specific anti-SARS-CoV-2 IgM immune response
- Qualitative test using Index , Single test, Ready to use (content is sealed strips), No extra reference required (Tests, Calibrator Controls in the kit), Samples : Serum, plasma (hep lit), Stability : 12 months
- Sens 100%, Spec 99.6% / PPV* 93,2%, NPV* 100%
| Weight | 1 kg |
|---|---|
| Certification | CE-IVD |
| Incoterms | EXW |
$360.00
VIDAS® SARS-COV-2 IgM (9COM) is an automated qualitative assay for use on the VIDAS® family of instruments, for the detection of immunoglobulin M (IgM) specific for SARS-CoV-2 in human serum or plasma (lithium heparin) using the ELFA (Enzyme Linked Fluorescent Assay) technique.This assay is intended for use as an aid to determine if individuals may have been exposed and infected by this virus and if they have mounted a specific anti-SARS-CoV-2 IgM immune response.In addition to viral load measured (e.g. real‑time RT‑PCR) in respiratory tract specimen, serology testing for specific immunoglobulins is another approach to identify individuals previously exposed to SARS‑CoV‑2.Indeed, most COVID‑19 patients had an antibody response at ten days or later after onset of the symptoms.This antibody response is characterized by the early rise of type M immunoglobulins (IgM), then followed by type G immunoglobulins (IgG).Furthermore, serum neutralization assays and virus culture have demonstrated the presence of neutralizing antibodies, that correlated to the presence of IgG recognizing the spike and the nucleoprotein of SARS‑CoV‑2.PRINCIPLEThe assay principle combines a two-step sandwich enzyme immunoassay method with a final fluorescence detection (ELFA).The single-use Solid Phase Receptacle (SPR) serves as the solid phase as well as the pipetting device. Reagents for the assay are ready-to-use and pre-dispensed in the sealed single-use reagent strips.All of the assay steps are performed automatically by the instrument. The reaction medium is cycled in and out of the SPR device several times.After the sample dilution step, the SARS‑CoV‑2 IgM are captured by recombinant SARS‑CoV‑2 antigen coated into the interior of the SPR device wall. Unbound components are eliminated during washing steps.During the second step, the IgM are specifically detected by anti-human IgM labeled with alkaline phosphatase. Unbound components are eliminated during washing steps.During the final detection step, the substrate (4-Methyl-umbelliferyl phosphate) is cycled in and out of the SPR device. The conjugate enzyme catalyzes the hydrolysis of this substrate into a fluorescent product (4-Methyl-umbelliferone), the fluorescence of which is measured at 450 nm.The intensity of the fluorescence is proportional to the level of antibody in the sample.At the end of the assay, the results are automatically calculated by the instrument according to the S1 standard stored in memory and a test value is obtained.The results can then be printed out
Additional information
| Weight | 1 kg |
|---|---|
| Certification | CE-IVD |
| Incoterms | EXW |
Vendor Information
- Store Name: bioMérieux
- Vendor: Lalla Haidara
-
Address:
376, chemin de l'Orme 69280 Marcy l'Etoile
69280 LYON
France - No ratings found yet!